Vacuum Dryer: The Complete Buying Guide In 2025
Vacuum Dryer: The Complete Buying Guide In 2025
Have you ever wondered how to sustain product quality when drying? You must be well aware of the demerits of conventional drying methods. Now, many of the old drying methods are obsolete. Because it can bring drying with compromised product quality.
A vacuum dryer is the solution to preserve the potency of your materials by subtly evaporating unwanted moisture. This blog is all about the vacuum dryer and extensive knowledge that you must know.
Whether you are a researcher, pharmacist, biotechnologist, technologist, or businessperson. This is the right place for you. Why? Here you can find A to Z information hooked to this equipment. Without wasting your time, let's jump into the main subject.
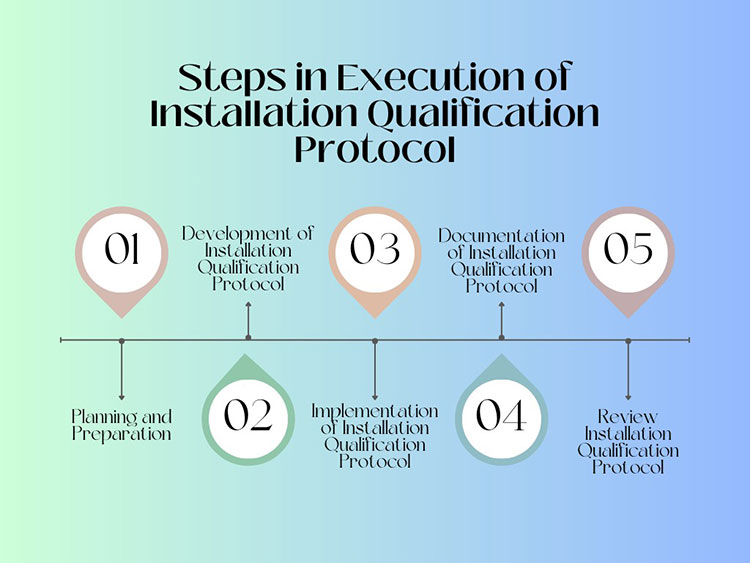
1.What is a vacuum dryer?

Vacuum Dryer
The vacuum dryer the suitable unit used for various batch processes to minimize the pressure of air. By reducing pressure directly lowers down the boiling point of the water.
Ultimately, the presence of moisture in the particles goes out and makes your content evenly dried. The machine is highly recommended for ingredients those are very sensitive to heat.
However, machine follows the subtle principles to maintain the potency of the products. Such as, inducing the heat which dried the content at a lower temperature. The vacuum itself is responsible for removal of air and moisture content. That’s why it is mainly responsible for the drying of heat sensitive materials.
2.What are the benefits of a vacuum dryer?
A vacuum dryer is considered as a quiet reliable unit for the drying process and has been utilized for a longer period of time. All due to their various benefits it is offering to their customers. That’s the reason, pharma, foods, biotechnology and various fields require it for attaining safe drying procedure. have a look at below discussed important advantages offered by a vacuum dryer.
Less Energy Expenditure More Drying

Energy Conservation- Picture Courtesy: Inspired Ingredients
Do you know that energy conservation is the biggest advantage of vacuum drying? This is true, you need less amount of energy which directly shortens your economic expenditures required for the drying process.
The vacuum dryer tends to make the drying process very fast therefore cutting down the time required for the drying of moisture present in the product.
Safe for your Ingredients

Preservation of ingredients
This is the second most important benefit of using a vacuum dryer where your ingredients are meant to be safe when it comes to dealing with this unit. This is pretty normal that some materials may be capable of experiencing high temperatures, but some do not.
This is nothing to worry about as a vacuum dryer is a reliable machine that helps in maintaining the integrity of the content without harming their qualities. That’s why pharma, foods, nutraceuticals, and many other facilities prefer vacuum dryer for drying procedures.
Secure for your Workers

Vacuum Dryer
If you see other kinds of dryers, they are constructed in a way that gives off fumes from vented parts. The generation of those particles in your environment makes the workers sick and also affects the working facilities. The vacuum dryer in this way is safer as it is capable of capturing the moisture in the collector with the help of a condenser and removed by using a vacuum pump.
High Productivity
Greater Production- Picture Courtesy: EnWave Corporation
As compared to traditional or direct modes of drying the contents, the vacuum dryer offers you greater production capacity. This is because the drying efficiency of the machine is so fast. It lowers the boiling point and induces the vacuum which offers a faster drying. You can enjoy maximum production capacity due to its rapid drying time.
Excellent Automation

Vacuum Dryer with Excellent Automation- Picture Courtesy: Amixon GmbH
The vacuum dryer is mainly designed with automatic keys which are features with auto-sensor, temperature regulator, induction of vacuum, drying time, etc.
With so many properties, you do not need to stand all the time to monitor the process. Similarly, as machine senses the temperatures, reduces the insight pressure, and develops a vacuum, with automatic time which is already fed in before starting the procedure.
So, with these features, you can only introduce the materials to be dried and start the machine, further operation will be performed by the machine automatically.
No More Chemical Reaction

Product’s Integrity- Picture Courtesy: Busch Australia
When you’re using a vacuum dryer, you will have greater control over the parameters inside the vacuum chamber. For example, you have controlled variables such as temperature and pressure. This means it can help in the prevention of chemical reactions happening inside. For example, there’s no chance of oxidation with certain medicines when encountered with a vacuum dryer.
Due to the vacuum, you can provide a gentle way of drying hence well-controlled process can be achieved without interrupting the content or substances with powdered texture.
3.What are the applications of vacuum dryer?
A vacuum dryer is widely utilized in the broad industrial sector as it offers you various distinguishing benefits. Let’s see the industrial applications of vacuum dryers.
Food Industry

Food industry- Picture Courtesy: Avid Armor
In the food industry, there are several goods prepared that are dried by vacuum dryer to preserve their potency freshness, effectiveness, and aroma. When you are subjecting them to a conventional dryer, there might be a greater possibility of damaging all the above properties of your food. Thus, vacuum dryer makes consumers appeal and efficacy of foods.
There are various nutritional ingredients such as spices, dried foods, snacks, coffee, powdered milk, tea, and various dried products such as pulses, grains, etc are dried by this unit which lowers the presence of water level by reducing the pressure and boiling point of water. Thus, more safe drying is achieved.
Pharmaceutical Industry

Pharmaceutical Industry- Scenesafe
When drugs have moisture content, it has more chance to be oxidized. In the pharmaceutical industry, no one can ignore the significance of vacuum dryers. Because it helps in the drying of drugs and medicines without affecting their therapeutic activities.
As medicines are mainly required for the treatment of various ailments therefore to prepare them under control conditions is mandatory and it must be promising in terms of treatment.
This is mainly used in the manufacturing of medicines to have them with stable shelf life, possessing greater integrity and high potency. For example, preparation of antibiotics, powder suspension, dry powders for topical preparations, etc.
Biotechnology

Biotechnology- Picture Courtesy: Boniik
This is a sensitive and complex field where advanced ways are applied to biological systems to bring new fruitful products. Various elements such as proteins, enzymes, microbiological assays, yeast, amino acids, and macroscopic as well as microscopic studies are carried out.
For this purpose, this sector is always installed with a vacuum dryer to achieve safe and desirable outcomes.
Research and Development

Research and development- Picture Courtesy: Vetoquinol Australia
In research and development, a vacuum dryer has been hooked to broad applications. It dries the assay powders and sensitive elements of research studies-controlled environment. Hence it supports the research assays by preserving the quality of the goods. For example, drying of raw materials, isolation of powders, antibiotics, various animal and herb samples, macroscopic and microscopic samples, etc to preserve them safely for a certain period of time.
Chemical Industry

Chemical Industry- picture Courtesy: EY
You can dry various chemicals whether it is heat-sensitive, non-heat-sensitive, reactive, or non-reactive by using a vacuum dryer. This machine ensures that chemicals are dried without interfering with their properties or affecting the characteristics of items. It ensures that unwanted chemical degradation or reaction is less likely to occur, and your product is kept preventive and can be released from the machine with the same intact properties.
Agriculture Industry

Agriculture Industry- Picture Courtesy: Crop Science Society of America
A vacuum dryer is used in the agricultural industry when drying the crops after washing and making them cleaned and entirely dried. This is required to increase the shelf life of agricultural products by maintaining their nutritional value and essential properties.
You can employ them in the preservation of seeds and may use them during breeding periods to get effective germination and extensive crop growth. Moreover, for the herb’s extraction in the agricultural field, a vacuum dryer is required to improve the extraction and good quality extraction.
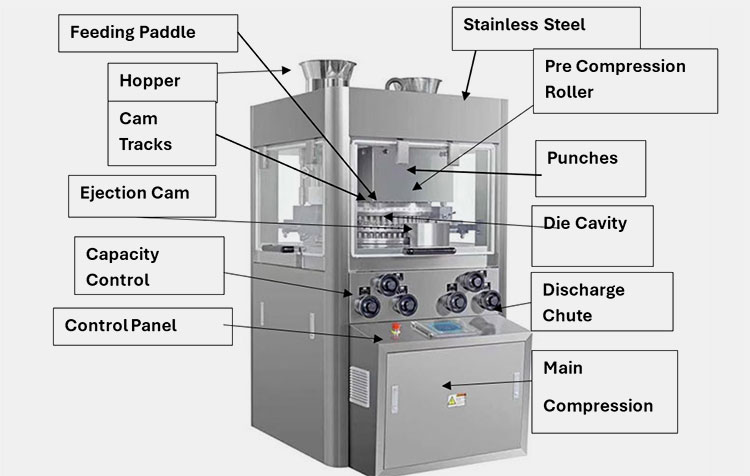
4.What is the construction of a vacuum dryer?

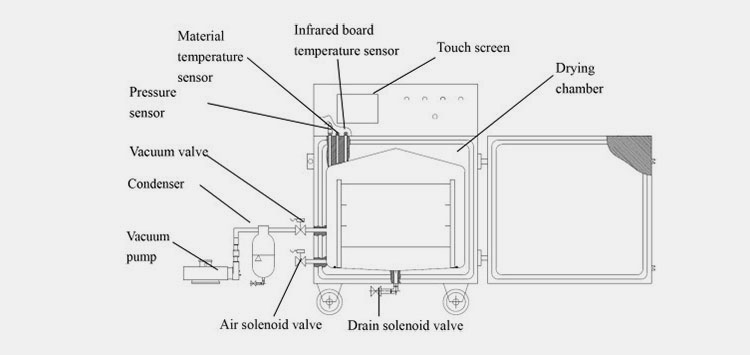
Parts of Vacuum Dryer
The parts and description are mentioned in the given table.
| Parts | Description |
| Door Gasket | A silicon or rubber-made door gasket is an important part of a vacuum dryer. The purpose of this specialized door is to attain an airtight sealing. When it is closed firmly, the possibility of achieving a vacuum is way faster. The outside air won’t be able to enter the unit so you can achieve the required drying rapidly. |
| Vacuum Gauge | The purpose of a vacuum gauge is to display the particular pressure gauge. You can easily determine the real-time measurements present inside the vacuum chamber. |
| Control Panel | You can control various characteristics such as vacuum pressure, drying time, temperature regulation, run time, troubleshooting, and other programmable settings are optimized by using a control panel. |
| Switches | Switches significantly enable you to operate the machine. They are simple and highly protective in terms of operation. You can turn the ON and OFF the machine with the help of switch regulations. |
| Valve | The regulation of airflow inside and outside the system is carried out by the help of valves. The proper working of valves ensures that the system is working effectively and the unit is safe without risks of contamination. |
| Observation Window | The glass window is mainly made up of transparent materials ensuring you monitor what’s going on inside. Here you don’t need to open and close the window again and again hence the vacuum condition becomes stable. |
| Shelf | You can place materials to be dried on the shelves. These are mainly perforated in design with stainless steel corrosion-free materials that ensure a safe and uniform airflow inside the chamber. Therefore, the number of items arranged on the trays or shelves receives maximum time and treatment to get sufficient air drying. |
| Handle | This is a pretty common part of the machine that allows you to open and close the gasket door conveniently. The handles are designed with a firm twisting mechanism that ensures an airtight closing of the door. |
5.What is the working of a vacuum dryer?
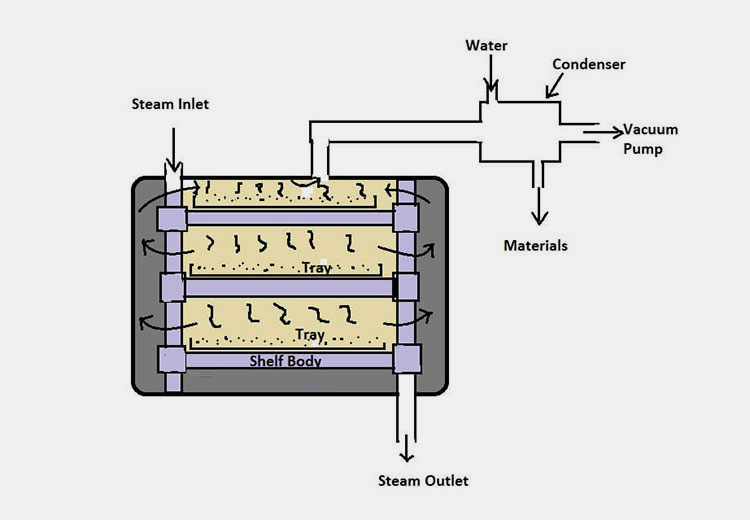
Working Principle of Vacuum Dryer
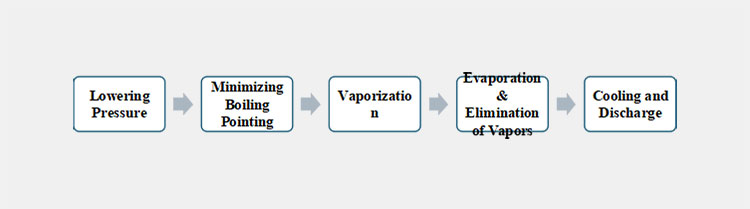
Process Steps of Vacuum Dryer
Step 1
As the chamber of the vacuum dryer is categorized with multiple shelves, therefore, you can simply place the sample to be dried over them. The basic working principle of the vacuum chamber is based on the induction of a vacuum. So, once you close the door tightly. The following process starts by simply starting the key on the control panel.
Step 2
The made up of vacuum dryer is associated with a vacuum pump which helps in the lowering of pressure inside. Also, the machine is integrated with a condenser which mainly supports your content from moisture as it evaporates effectively.
The typical range for vacuum pressure lies between 30 to 60 Kpa. This is mainly achieved by the utilization of vacuum pumps.
Moreover, the steam inside the chamber is evenly distributed via conduction. In this state, the water molecules evaporate quickly. The optimum temperature used is around 25-30°C.
Upon generation of vacuum, the process of vaporization becomes faster with simultaneous heat conduction which helps in the evaporation of water or moisture content. They’re mainly removed from the vacuum pump to avoid being transformed back thus you can get sufficient drying.
Step 3
After vacuum drying, your materials are cooled down. After achieving certain stabilization, you can carefully collect the materials from the shelves.
6.What is the classification of a vacuum dryer?
There are certain types of vacuum dryer. Let’s see them below:
Benchtop Vacuum Dryer

Benchtop vacuum dryer- Picture Courtesy: NBchao.com
This is commonly known as a lab vacuum dryer or static vacuum dryer. In this machine, uniform heat conductivity is generated and supplied over various shelves.
The process of drying for thick or viscous materials is a bit slow; it is because vacuum drying is not accompanied by agitation.
There are various examples of benchtop or lab vacuum dryer. For example, tray lab vacuum dryer, shelf vacuum dryer, and microwave vacuum dryer.
Tumbling Vacuum Dryer

AIPAK Tumbling Vacuum Dryer
It is another primary kind of vacuum dryer. Since it employs the rotating or agitating drum or vessel, it is also called a rotary vacuum dryer. The materials rotate in the vessel in the vacuum condition. The products are continuously exposed to high temperatures due to the rotation of the vessel, leading to faster drying times.
Variances in moisture retention are minimized due to constant rotational motion, which improves uniform material drying. Furthermore, it can treat bulk volumes of products and thus is more suited to mass-scale manufacturing. But it is less ideal for processing fragile products, as it degrades them. Its subcategories are:
Double Cone Rotary Vacuum Dryer

Allpack Double Cone Rotary Vacuum Dryer
A double-cone rotary vacuum dryer is an ideal solution for pulp, viscous, and thermolabile content which is mainly dried under low temperatures. The machine is capable of dealing with high-capacity content and materials prone get oxidized rapidly. This controlled unit, it can handle products without harming the integrity of raw materials.
Vacuum Pan Dryer

Vacuum Pan Dryer- Picture Courtesy: Heinkel
It is the form of a vacuum dryer in which heat-sensitive, pasty, and viscous products are heated more quickly with the aid of axial and radial agitators. It consists of a circular pan that offers a flat surface for even distribution of materials. This pan can be turned or rotated to speed up the drying process.
Vacuum pan dryer has the shortest drying cycle and produces even mixing without any lumps or balls due to its constant agitating movement.
Spherical Vacuum Dryer

Spherical vacuum dryer- Picture Courtesy: gmmpfaudler
The machine is designed with a hollow heated vessel with a jacketed wall. The presence of a rotating agitator ensures materials' uniform drying throughout the process. The spherical disc helps in the quick opening and closing of the system of charge and discharging of content.
Conical Vacuum Dryer

Conical Vacuum Dryer- Picture Courtesy: Hosokawa micron
The conical vacuum dryer mainly utilizes convection mixing for the drying. The constant rotation of the mixer ensures convective forces inside the conical vessel. For the drying procedure heat energy is required which is transferred into the vessel via a jacketed vessel that is induced by either hot air, water, or other media.
The presence of a vacuum ensures the elimination of vapors at a very low temperature that’s why it is known as an ideal solution for thermolabile products.
Horizontal Vacuum Dryer

Vacuum Belt Dryer- Picture Courtesy: Bucher Unipektin
This vacuum dryer is equipped with a conveyor belt that aids in homogenous drying. This belt conveys samples through various heating zones and a cooling zone in the vacuum chamber. The heat is administered from above or below the belt. The materials are placed on the parallel running belts. It has a design like an industrial boiler or a silo.
The vacuum belts have a direct connection with the heating and cooling plates, so, they directly transfer heat for drying products. Its drying temperature is about 35℃-90℃ and vacuum pressure is approximately 10mbar to around 80mbar.
Vertical Vacuum Dryer

Vertical Vacuum Dryer- Picture Courtesy: HEINKEL Group
It is identical in construction to a rotary vacuum dryer, except it has a vertical configuration with a conical bottom structure. This part is advantageous because of its minimal footprint, complete product discharge with lesser holdup, and easy cleanability. It also has an agitator that thoroughly mixes and dries while preventing lumping and caking.
It can accommodate about 25-13,000 liters of product while its drying temperature is about -10 to 150°C.
7.Can a vacuum dryer be suitable for dusty or hygroscopic materials?

Hygroscopic Materials
Yes, why not? A vacuum dryer is considered to be the safest unit for the drying of content those are hygroscopic or dusty in nature. Conversely, using hot air dry can affect the integrity of thermolabile and hygroscopic materials.
Therefore, a vacuum dryer is suitable as it mainly works under low pressure and reduced temperature which is a good option for heat-sensitive and hygroscopic goods. A vacuum dryer is demonstrated to show a greater rehydration rate than a conventional way of drying.
Therefore, this is the only option for manufacturing and other fields to use for the drying of hygroscopic materials, glutinous, or lumpy materials.
8.Is a vacuum dryer the same as a freeze dryer?

Vacuum Dryer- Picture Courtesy: Green Thumb Depot
Vacuum dryers and freeze dryers are different from each other. Generally, vacuum dryers and freeze dryers are applied in de-moisturizing processes within industries, but they operate on fundamentally different principles. Knowing the difference between them will guide you on which of the two is best for your case.
Here is a short comparison of vacuum dryers versus freeze dryers on key aspects:
Drying Mechanism

Mechanism
| Vacuum Dryer | Freeze Dryer |
| It operates on the principle of a low-pressure environment that creates a reduced boiling point for water. | It uses the lyophilization process. Material is frozen, then pressure is decreased enough that the ice sublimates directly into vapor without going through the liquid phase. |
Temperature and Pressure

Freeze Dryer (Temperature-40 to -80) - Picture Courtesy: Barnalab
| Vacuum Dryer | Freeze Dryer |
| It is mainly operated at mild temperature range i.e. 40°C - 100°C. | It works at very low temperatures, sometimes between -40 and -80 degrees Celsius, and very high vacuum conditions. |
Drying Time

Shorter Drying Time in Vacuum Dryer-Picture Courtesy: Waldner
| Vacuum Dryer | Freeze Dryer |
| Vacuum dryer has a relatively fast drying cycle, in the order of minutes or hours depending on the material and the level of moisture. | Freeze dryer requires hours to days, and every step includes freezing, sublimation, and the final drying, which require careful handling. |
Energy Consumption

Freeze Dryer- Energy Intensive – Picture Courtesy: Senieer
| Vacuum Dryer | Freeze Dryer |
| It is regarded as more energy-efficient energy because it operates at intermediate temperatures and needs only a vacuum pump to maintain reduced pressure. | Freeze drying requires much energy as it involves working under low temperatures and maintaining refrigeration and a vacuum. |
Cost and Machinery

High-tech and Expensive Requirement for Freeze Dryer -Picture Courtesy: Industrial-Integration
| Vacuum Dryer | Freeze Dryer |
| The process uses less and inexpensive equipment, making vacuum drying inexpensive. | Freeze drying requires sophisticated, high-cost equipment due to refrigeration systems, vacuum pumps, and special chambers. |
9.Does the Vacuum Dryer Follow a Batch or Continuous Process?
Today, vacuum dryer are available in a broad category that follows both processes such as batch and continuous process.

Vacuum dryer- Picture Courtesy: Tefic. biotech
Nevertheless, lab vacuum dryers or many benchtop vacuum dryers are designed in a way that follows a batch process. This means you can load samples for treatment and receive them once cooled and dried. For example, double-cone rotary vacuum dryers, conical, vertical vacuum dryers, etc.
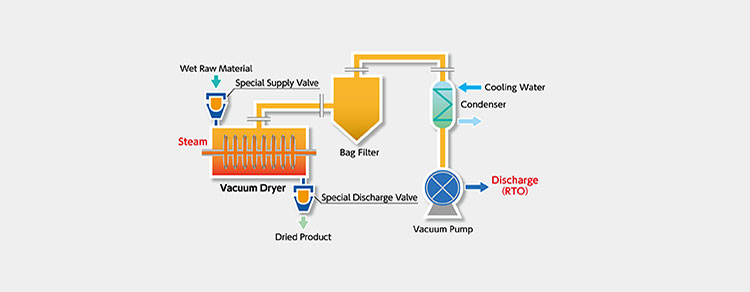
Design flow chat of continuous vacuum dryer- Picture Courtesy: nara.co.japan
However, newly developed innovation ensures a continuous process by supplying and discharging the sample with the help of supply valves making it possible to achieve a constant and continuous vacuum drying process.
10.What are the technical problems associated with vacuum dryer? How to solve them?
Vacuum dryers are extensively employed in numerous industries for effective drying of various materials and products. But have you ever come across some technical issues with their performance? Let’s dive into some common snags associated with vacuums and how you can deal with them.
Having Trouble with Insufficient Drying Efficiency

Vacuum Pump Maintenance
| Issue | Troubleshooting |
| If there isn’t enough vacuum or less efficient heat transfer, have you observed the drying process dragging on? These two considerations can slow down the performance and the productive moisture removal. | To keep things running smoothly, make sure that:
The heating components are in working order. The vacuum pump has to be sized appropriately and serviced frequently. Periodic calibration of the system for both temperature and pressure. |
Coping with Equipment Contamination

Contamination Control- Picture Courtesy: Griffin Machinery
| Issue | Troubleshooting |
| Lubricants, residues, or even dust from the vacuum chamber can sneak in and deteriorate the quality standard of your product. | To keep your vacuum dryer in excellent order, keep these points in consideration:
The materials used are repellent to contamination. Keep an eye on vacuum chamber leakage. Look over for residue and contamination regularly. Let’s have a satisfactory cleaning maintenance routine in place. |
Lack of Exhaust Control

Valve for Exhaust Control- Picture Courtesy: Amixon GmbH
| Issue | Troubleshooting |
| Have you ever noticed that introducing dry air or nitrogen during the drying process might impact fine powder? If the exhaust flow is inadequate, it can lead to powder washing away and particle loss. | Using a regulator or valve to accurately control the exhaust stream can really help manage problems that arise with the introduction of nitrogen or dry air. It’s crucial to pay attention to balancing water vapor elimination without losing powder. |
Noticed Vacuum Level Fluctuations

Vacuum Stability- Picture Courtesy: MJ Dryer
| Issue | Troubleshooting |
| Have you ever encountered rapid variations in a vacuum? It may lead to the powder material escaping into the atmosphere. | Keep in mind that the vacuum level needs to be adjusted gradually. Rapid changes can affect the stability of particles and create dust. |
Facing Temperature Control Issues

Temperature control system inspection- Picture Courtesy: BINDER GmbH
| Issue | Troubleshooting |
| This is pretty common that utmost alterations in drying temperature generate excessive heating, In fact, affects the quality of the product. | Ensure these key aspects:
Frequent calibration of sensors. Uniform heat distribution—temperature profiling. |
Spotting Vacuum Leakages

Leak Proof Vacuum System- Picture Courtesy: Lodha Pharma
| Issue | Troubleshooting |
| If you’re experiencing low-pressure conditions owing to a leak in your vacuum system? Let’s find some quick solution. | Pay attention to:
Manage systematic inspection of seals, gaskets, and connections for any wear and damage. A vacuum leak detector helps you to detect repair leaks on time. |
Struggling with High Energy Consumption

Energy-Efficient Vacuum System- Picture Courtesy: DCM Delta
| Issue | Troubleshooting |
| If you are experiencing incompetent operation and unsatisfactory insulation, this can contribute to enormous energy usage and operational costs. | The possible strategies include:
Optimize the parameters of the drying process for heat loss reduction and to improve insulation. Moreover, utilize energy-saving components. |
Mechanical Failures

Schedule maintenance- Picture Courtesy: MJ Dyers
| Issue | Troubleshooting |
| You may experience a mechanical breakdown decline in efficiency attributed to wear and tear in moving parts of vacuum pumps and dryers. This is particularly concerning for essential components such as seals. | You must consider regular maintenance to identify early wear in parts. This proactive approach helps in sudden mechanical failure and smooth operation. For example:
Incorporate lubrication. Part replacement. Regular inspection. |
Conclusion
A vacuum dryer is a one-stop solution for drying thermolabile and hygroscopic content in the safest manner. That’s the reason it is used for not only medicinal purposes but also in various fields of manufacturing sectors discussed above. This is a profitable unit in a various way such as energy conservation, saving your time, saving product loss, and much more. Willing to purchase a vacuum dryer? Why not contact our team? Why us? We are the experts in offering a series of reliable and economical machines that firmly adhere to FDA regulations. Send a Quote to begin the journey of success with us.
Don't forget to share this post!
CONTACT US
Tell us your raw material and project budget to get quotations within 24 hours.
WhatsApp Us: +86 181 6426 8586
The Buyer's Guide
 Tell us your material or budget, we'll reply you ASAP within 24 hours
Tell us your material or budget, we'll reply you ASAP within 24 hours
Vacuum Dryer: The Complete Buying Guide In 2025 Read More »























 Tell us your material or budget, we'll reply you ASAP within 24 hours
Tell us your material or budget, we'll reply you ASAP within 24 hours